Anutosh Chakraborty, Ph.D.
Associate Professor
Pharmacology and Physiology
Education
Ph.D. Indian Institute of Chemical Biology, Jadavpur, India 2005
"Chaperonic Function of a Cyclophilin Homolog from Leishmania donovani"
M.Sc. Burdwan University, India 1997
Zoology (First class)
B.Sc. Burdwan University, India 1995
Zoology major, Chemistry, Botany (First Class)
Previous Positions
Associate Professor
St. Louis University School of Medicine, St. Louis, MO. Aug 2017-
Associate Professor
Scripps Research Institute, Jupiter, Florida. June 2017-July 2017
Assistant Professor
The Scripps Research Institute, Jupiter, Florida. 2012-June 2017
Research Associate and Instructor
Johns Hopkins Medical School, Baltimore, Maryland 2008-2012
Postdoctoral fellow
Johns Hopkins Medical School, Baltimore, Maryland 2005-2008
Research Trainee
Johns Hopkins Medical School, Baltimore, Maryland 2004-2005
Research
Obesity and type-2 diabetes mellitus (T2DM) significantly increase the risk of non-alcoholic fatty liver disease/steatohepatitis (NAFLD/NASH), which can also lead to liver cirrhosis and hepatocellular carcinoma. No FDA-approved drugs are available to treat NASH. Limited success in treating diseases is partly due to inadequate understanding of the cellular mechanisms that cause these illnesses. Although majority of the FDA-approved drugs work via targeting proteins, in vivo functions of ~10-15% annotated mammalian proteins are unknown. Using diverse approaches shown below, my laboratory focuses on identifying and characterizing novel proteins in cell metabolism and signaling. We also identify targets and regulators of these proteins and test their therapeutic potential in metabolic diseases.
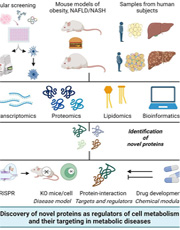
The major ongoing projects in the lab are described below.
IP6K1 is a novel target in obesity, T2DM and NASH. Our group established that disruption of the signaling inositol pyrophosphate (5-IP7)
biosynthetic enzyme IP6K1 ameliorates metabolic diseases. Ip6k1 knockout mice are protected from diet-induced obesity (DIO), hyperinsulinemia, hyperglycemia,
and NAFLD/NASH due to increased energy expenditure and insulin sensitivity. Moreover,
we demonstrated that the IP6K pathway can be pharmacologically targeted in metabolic
diseases. Thus, IP6K1 inhibition should have pleiotropic benefits on human metabolic
health. Structure-activity relationship (SAR), pharmacokinetic and cellular studies
are being conducted to develop orally available IP6K1-selective analogs. Hopefully,
IP6K inhibitors will have therapeutic values in obesity, T2DM, NAFLD/NASH, osteoporosis,
and aging.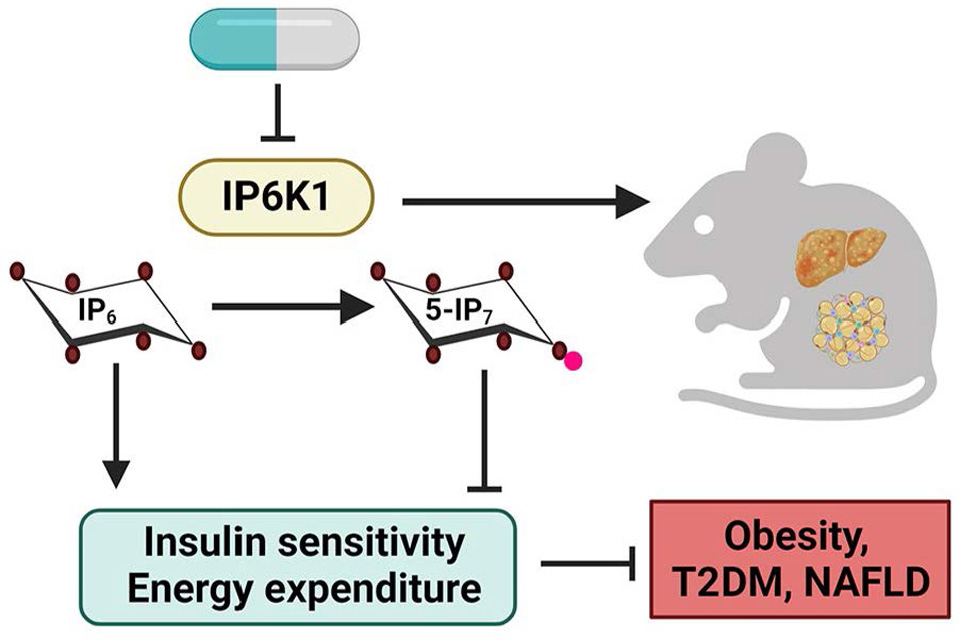
Deciphering metabolic functions of the E3 ligase Ube4A. E3 ubiquitin ligases control diverse cellular functions by ubiquitination-mediated
regulation of protein targets, and thus their functional aberration is associated
with many diseases. RING-, HECT- and RBR-domain containing E3 ligases are the most
studied in mammals. In addition, a distinct family of E3 ligases exist that contain
the U-box domain, resembling RING but lacking the Zn2+-coordinating residues. Mammals only have seven U-box ligases including Ube4A. The
in vivo function of Ube4A is unknown. By generating whole-body Ube4A knockout (UKO) mice by the CRISPR/Cas9 method, we discovered that Ube4A deletion augments DIO, insulin resistance, hyperinsulinemia and NAFLD. Studies are
ongoing to 1) determine cell-specific role of Ube4A in metabolic diseases; 2) identifying
targets and regulators of Ube4A and deciphering their role in these diseases and 3)
development of Ube4A activator compounds for the treatment of obesity and NAFLD/NASH. 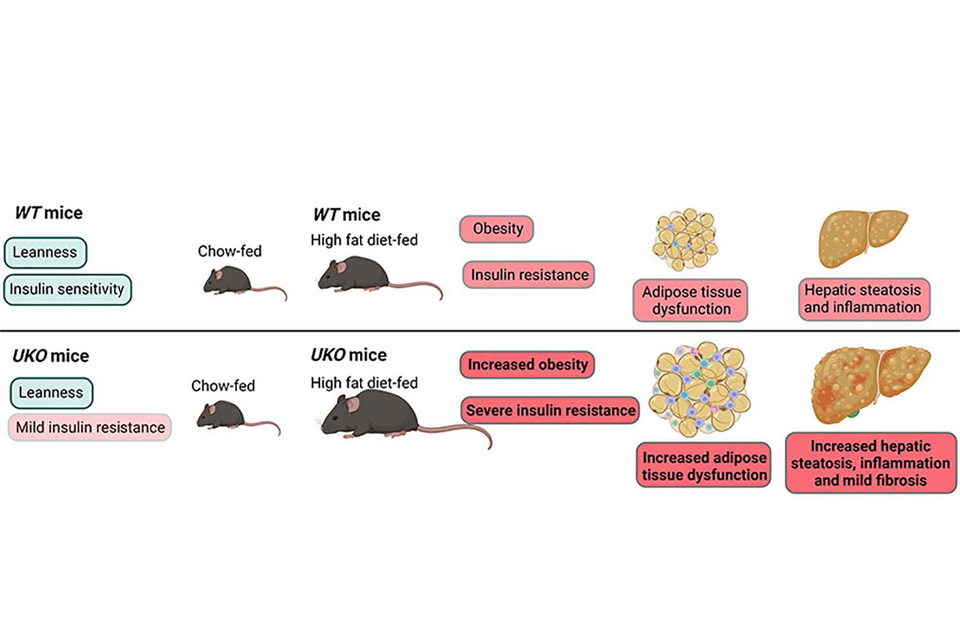
Identifying novel regulators of obesity, NAFLD/NASH, and drug development. Our studies in mice and human patients identified many uncharacterized proteins that
are altered in NAFLD/NASH. Studies are ongoing to determine their functions in this
disease. Moreover, cellular assays are being used to screen chemical libraries to
identify compounds that enhance metabolism and insulin sensitivity and reduce inflammation
and collagen formation. We have identified several lead compounds, the efficacy of
which are being tested in mouse models of NASH. Thus, diverse approaches taken in
my laboratory will not only help to understand functions of the hitherto unappreciated
proteome but also develop new therapeutic strategies for the treatment of metabolic
diseases. Figures are generated using the Biorender software.
Professional Experience
Mentoring Experience
Postdoctoral fellows
Vineet Babu: Sept 2023 -
Sandip Mukherjee: Oct 2018 – May 2022 (Current position: Staff Scientist, Division of Geriatrics and Nutritional Science, Washington University, Saint Louis, MO)
Qingzhang Zhu: March 2013 - January 2017 (Current position: Instructor, Internal Medicine, UT Southwestern Medical Center, TX)
Sarbani Ghoshal: May 2013 - August 2016 (Current position: Faculty, City University of NY)
Graduate students
Eliwaza Nao Msengi (July 2018 – May 2023): Thesis title: Defining the mechanistic role of the U-box E3 ligase Ube4A in metabolic health and diseases.
Jake Haubner (July 2019 -)
Master’s thesis
Darlena Henderson: 2013. Thesis title: Inositol Hexakisphosphate Kinase-1 stabilizes the lipolytic regulator protein, perilipin1.
Hector Mora: 2015. Thesis title: Inositol pyrophosphate and ubiquitin-proteasomal pathways regulate cell signaling and metabolism.
Research Technician and lab manager
Molee Chakraborty: May 2017 –
Ana Rodrigues: February 2013-March 2014
Publications and Media Placements
Commentaries / Media
Dr. Chakraborty- Awarded 4 Year RO1 for NAFLD/NASH
Scripps Florida Scientists Take Aim at Obesity-Linked Protein
https://www.eurekalert.org/pub_releases/2016-10/sri-sfs101216.php
https://www.sciencedaily.com/releases/2016/10/161012095617.htm
Scripps Scientists Win $2.2 Million to Expand Study of Innovative Obesity Therapy
https://www.scripps.edu/news/press/2015/20150527chakraborty.html
Obesity and diabetes: Helping PIP3 to get in on the Akt [https://www.lipidmaps.org/update/2011/110101/full/lipidmaps.2010.39.html]
https://www.nature.com/scibx/journal/v4/n1/full/scibx.2011.1.html
Publications
Major publications
- Mukherjee S#, Chakraborty M#, Msengi EN#, Haubner J, Zhang J, Jellinek M, Carlson H, Pyles K, Ulmasov B, Lutkewitte AJ, Carpenter D, McCommis KS, Ford DA, Finck BN, Neuschwander-Tetri BA, Chakraborty A*. Ube4A maintains metabolic homeostasis and facilitates insulin signaling in vivo. Mol Metab, 2023, 75:101767.
- Mukherjee S, Chakraborty M, Haubner J, Ernst G, Carpenter D, Barrow JC, and Chakraborty A*. The IP6K inhibitor LI-2242 ameliorates metabolic dysfunction in diet-induced obese mice. Biomolecules, 2023, 13
- Zhou Y, Mukherjee S, Huang D, Chakraborty M, Gu C, Zong G, Stashko MA, Frye SV, Pearce KH, Shears SB, Chakraborty A*, Wang H*, Xiaodong Wang X*. Development of novel IP6K inhibitors for the treatment of obesity and obesity-induced metabolic dysfunctions. Med. Chem. 2022, 65, 6869-6887.
- Mukherjee S, Chakraborty M, Ulmasov B, McCommis K, Zhang J, Carpenter D, Msengi EN, Haubner J, Guo C, Pike DP, Ghoshal S, Ford DA, Neuschwander-Tetri BA, Chakraborty A*. Pleiotropic actions of IP6K1 mediate hepatic metabolic dysfunction to promote nonalcoholic fatty liver disease and steatohepatitis. Mol Metab., 2021, 54, 101364
- Mukherjee S, Haubner J, Chakraborty A*. Targeting the Inositol Pyrophosphate Biosynthetic Enzymes in Metabolic Diseases. Molecules., 2020, 25, pii: E1403
- Chakraborty A*. The inositol pyrophosphate pathway in health and diseases. Biol Rev Camb Philos Soc., 2018, 93, 1203-1227. Review.
- Zhu Q, Ghoshal S, Tyagi R and Chakraborty A*. Global IP6K1 deletion enhances temperature modulated energy expenditure which reduces carbohydrate and fat induced weight gain. Metab., 2016, 6, 73-85.
- Zhu Q, Ghoshal S, Rodrigues A, Gao S, Asteian A, Kamenecka TM, Barrow JC and Chakraborty A*. Adipocyte-specific IP6K1 deletion reduces diet-induced obesity by enhancing AMPK mediated thermogenesis. Clin. Invest., 2016, 126, 4273-4288
- Ghoshal S, Zhu Q, Asteian A, Lin H, Xu H, Ernst G, Barrow JC, Xu B, Cameron MD, Kamenecka TM and Chakraborty A*. TNP [N2-(m-Trifluorobenzyl), N6-(p-nitrobenzyl)purine] ameliorates diet induced obesity and insulin resistance via inhibition of the IP6K1 pathway. Metab., (2016), 5, 903-17.
- Chakraborty A, Koldobskiy MA, Bello NT, Maxwell M, Potter JJ, Juluri KR, Maag D, Kim S, Huang AS, Dailey MJ, Saleh M, Snowman AM, Moran TH, Mezey E and Snyder SH. Inositol pyrophosphates inhibit Akt signaling, thereby regulating insulin sensitivity and weight gain. , (2010), 143, 897-910.
Grants
RESEARCH SUPPORT
Active
R01 DK132162 (2023-2026, $2,133,260.00)Source/Role: NIH/NIDDK (PI)
Title: Determining hepatocyte-specific mechanisms by which Ube4A regulates NAFLD/NASH
T32 GM141602 (2023-2027)
Source/Role: NIH/NIGMS (MPI)
Title: Pharmacological Sciences Training Grant
Saint Louis University Start-up fund (2017 -)
Completed
R01 DK103746-01: 04/11/15 – 03/31/21Source / Role: NIDDK / PI
Title: Inositol hexakisphosphate kinase-1 As a Novel Target in Obesity
R21 NS108138-01: 09/01/2018 - 08/31/2020
Source / Role: NINDS / CO-PI
Title: Role of adropin in maintaining cognitive function
Research Opportunity Fund
Source/Role: Saint Louis University / PI
Title: Testing efficacy of IP6K inhibitors in vivo
Liver-Center Grant (2018)
Source/Role: Saint Louis University / PI
Title: Determine functions of novel proteins that link ER-stress to NAFLD
President Research Fund (2019)
Source/Role: Saint Louis University / PI
Title: Determine mechanisms that regulate the obesogenic protein IP6K1
Honors and Awards
2023: Outstanding Teacher Award, Graduate Program, Saint Louis University
2023: Editorial board member: The Journal of Biological Chemistry (JBC)
2023: Ad Hoc reviewer: NIH HBPP study section
2022: Ad Hoc reviewer: NIH BMDM study section
2021: Ad Hoc reviewer: NIH HSDO study section
2019: Session Chair: Keystone Symposium on Integrated Pathways of Disease in NASH/NAFLD in Santa Fe
2017: Outstanding Reviewer, Journal of Molecular Biology (JMB)
2017: Outstanding Reviewer, Cellular Signaling
2016: Recognized reviewer, Journal of Molecular Biology (JMB)
1999: Research Grant and Fellowship by the CSIR / UGC, India
1999: Ranked 2nd (99.87 percentile) at the Graduate Aptitude Test in Engineering, conducted nationally by the Indian Institute of Technology (IIT)
1998: Lectureship by the CSIR / UGC, India
Professional Organizations and Associations
The Obesity Society (TOS)
American Diabetic Association (ADA)
Missouri Physiological Society (MOPHYS)
American Association for the Study of Liver Diseases (AASLD)